 Padmanabhan Ramnarayan, MD Alvin Richards-Belle, BSc Laura Drikite, MSc Michelle Saull, BSc Izabella Orzechowska, MSc Robert Darnell, MA Zia Sadique, PhD Julie Lester, BA Kevin P. Morris, MD Lyvonne N. Tume, PhD Peter J. Davis, MBChB Mark J. Peters, PhD Richard G. Feltbower, PhD Richard Grieve, PhD Karen Thomas, MSc Paul R. Mouncey, MSc David A. Harrison, PhD Kathryn M. Rowan, PhD FIRST-ABC Step-Down RCT Investigators and the Paediatric Critical Care Society Study Group Nazima Pathan Esther Daubney Deborah White Nayan Shetty Dawn Jones Laura Rad Laura O'Malley Kevin Morris Sarah Fox Carly Tooke Afeda Mohamed Ali Peter Davis Helen Marley Rebecca Lean Laura Dodge Angela Aramburo Laura Alcantara Laura Tos Helena Sampaio Siva Oruganti Susan Bowes Awen Hughes Mark J Peters Lauran O'Neill Holly Belfield Samiran Ray Rohit Saxena Helen Vander Johnson Tara McHugh Gareth Jones David Armstrong Laura Fraser Margrethe Van Dijke Ian Piper Jon Lillie Paul A Wellman Aleksandra Williams Tabitha Craen Joanne Perkins Christine Mackerness Aravind Kashyap Lindsay Cooper Angela Lawton Lynda Verhulst Akash Deep Ivan C Caro Eniola Nsirim Samira N Vahid Bedangshu Saikia Rekha Patel Graham Mason Claire Jennings Rebecca Marshall Danielle Pask Avishay Sarfatti Zoe Oliver Katie Wingfield Sophie Herrington Caterina Silvestre Laura Anderson Maria Saxton Helen Fazackerley Naomi Edmonds Natasha Thorn Nosheen Khalid Hafiza Khatun Anton Mayer Alex Howlett Jade Bryant Ahmed Osman Amber Cook Lorena Caruana Phillipa C Thomas Nicholas J Prince Joana G de Queiroz Elena Maccarcari Montserrat R Foguet Rebecca Mitting Sarah Darnell David Inwald Sam Peters Lorna Miller Stefan Sprinckmoller Abby Koelewyn Roger Parslow Summary of Adverse Events and Serious Adverse Events Baseline Characteristics in All Randomized and Consented Children Irrespective of Whether Respiratory Support Was Started or NotĮTable 10. Timing and Reasons for Treatment Failure (Switch/Escalation Events) in Children Who Started the Allocated TreatmentĮTable 9. Devices and Interfaces Used in Children Who Started the Allocated TreatmentĮTable 7. 
Adherence With Trial Algorithms in Children Who Started the Allocated TreatmentĮTable 6. Baseline Characteristics in the Per-Protocol PopulationĮTable 5. Additional Baseline Characteristics, Including Physiological Variables Split by Child on/Not on Non-invasive Respiratory Support at Randomization in the Primary Analysis SetĮTable 4. 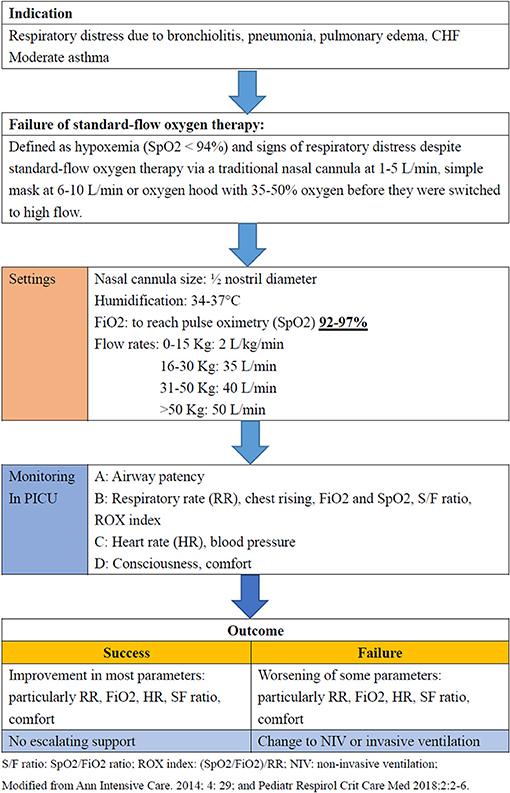
Characteristics of Participating UK National Health Service Critical Care UnitsĮTable 3. Patient Data Collection ScheduleĮTable 2. Time to Liberation From Respiratory Support – Post-Hoc Sensitivity Analysis in All Randomized Children Including Those Who Were Not Started on Respiratory SupportĮTable 1. Respiratory Support Treatments Provided Over Time to Children in the Primary Analysis SetĮFigure 10. Breakdown of the Time to Liberation From Respiratory Support by Occurrence of Treatment Failure in Children Who Started the Allocated TreatmentĮFigure 9. Clinical Management of Trial PatientsĮFigure 8. CPAP Pressures During the First Six Hours of TreatmentĮFigure 7. HFNC Flow Rates During the First Six Hours of TreatmentĮFigure 6. Screening, Randomization and Follow-up in the Per-Protocol PopulationĮFigure 5. Actual Versus Anticipated Patient RandomizationĮFigure 4. Trial Algorithm for the Delivery of Continuous Positive Airway Pressure (CPAP)ĮFigure 3.  
Trial Algorithm for the Delivery of High Flow Nasal Cannula (HFNC)ĮFigure 2. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
